
The basic thought of what is molecular orbitals can be the organized combinations of the atomic orbitals according to the symmetry of the molecules and the characteristics of atoms. The reason for the unexpected stability of organic compounds in an oxygen atmosphere is that virtually all organic compounds, as well as H 2O, CO 2, and N 2, have only paired electrons, whereas oxygen has two unpaired electrons. Since each hydrogen atom has a single 1s atomic orbital for its electron, the overlap of these two atomic orbitals forms a bond. Just like the atomic orbitals, molecular orbitals(MO) are used to describe the bonding in molecules by applying the group theory. Draw the molecular orbital diagram of dioxygen and calculate bond order. We again fill the orbitals according to Hund’s rules. To obtain the molecular orbital energy-level diagram for O 2, we need to place 12 valence electrons (6 from each O atom) in the energy-level diagram shown in Figure 9.10.1. Reason : Electrons are removed from the antibonding molecular orbital from H2. The bond length in the oxygen species can be explained by the positions of the electrons in molecular orbital theory. Fortunately for us, however, this reaction is very, very slow. In the molecular orbital diagram for the molecular ion, N + 2, the number of electrons in the 2p molecular orbital is : Q. Demonstrate how Hü ckels theory approximates the full molecular orbital picture of molecules by treating the (sigma)-bonding and (pi)-bonding networks independently. These are sometimes denoted, in MO diagrams like the one below, with the Greek letter psi ( ) instead of. Because Earth’s atmosphere contains 20% oxygen, all organic compounds, including those that compose our body tissues, should react rapidly with air to form H 2O, CO 2, and N 2 in an exothermic reaction. According to MO theory, the two atomic 2 pz orbitals combine to form two pi () molecular orbitals, one a low-energy bonding orbital and one a high-energy -star () anti-bonding molecular orbital. Full video can be found at The magnetic properties of O 2 are not just a laboratory curiosity they are absolutely crucial to the existence of life. In the molecular orbital diagram for the molecular ion, N2+, the. As atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of molecular orbitals. Consequently, it is attracted into a magnetic field, which allows it to remain suspended between the poles of a powerful magnet until it evaporates. Make a molecular orbital diagram for SO2 using group theory Consider the Molecular Orbital Diagram of N2+ and answer the following questions: Write a correct molecular orbital diagram for each of the following. The potential energy curve for the H2 molecule as a function of internuclear distance is . MO Diagram - A molecular orbital diagram, also known as a MO diagram, is a qualitative descriptive tool used to explain chemical bonding in molecules using molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular. 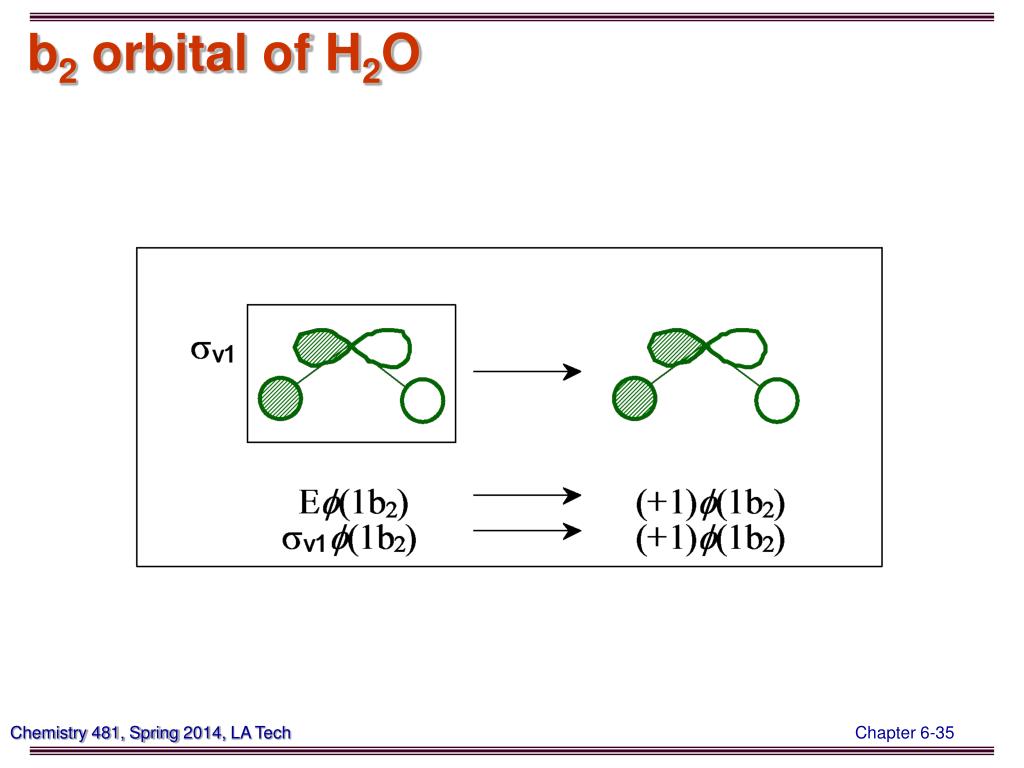
\): Liquid O 2 Suspended between the Poles of a Magnet.Because the O 2 molecule has two unpaired electrons, it is paramagnetic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
